
However, reappearance of almost 50% of these epitopes was observed within 48 h incubation at 37 oC ( 2). Enzymatic removal of the terminal galactose (Gal) from a-gal epitopes on porcine endothelial cell membranes by a-galactosidase prevented anti-Gal binding. This epitope is absent in humans and is an obstacle in xenotransplantation because the natural anti-Gal antibody produced in large amounts in humans binds to xenografts presenting it and causes their rejection. The a-gal epitope is abundant in non-primate mammals, prosimians and New-World monkeys. The kinetics of ABO antigen reappearance on nucleated cells can be inferred from studies on reappearance of the a-gal epitope (Gala1-3Galb1-4GlcNAc-R) which is a glycan closely resembling ABO antigens. Therefore, removal of carbohydrate-antigens from the membranes of nucleated cells by glycosidases is followed by reappearance of these antigens. Nucleated cell membranes constantly undergo turnover, resulting in de-novo appearance of glycolipids and glycoproteins with linked carbohydrate chains (glycans) synthesized within the Golgi apparatus by glycosyltransferases, including ABO-transferases. Reappearance of enzymatically removed incompatible carbohydrate-antigens such as ABO antigens from human allografts may be a major obstacle to the success of grafting, if no additional steps are taken for preventing rejection by the incompatible antibodies. However, they indicated that such reappearance was not observes for several hours after the enzymatic treatment. 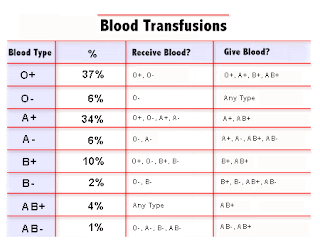
The authors recognized the study limitations regarding the possible reappearance (re-expression) of the A-antigen, because of cellular glycosyltransferases activity. Wang et al indicate that enzymatic conversion of blood type-A into O lungs will enable the use of such lungs as allografts in ABO incompatible recipients, without the risk of graft rejection by anti-blood group-A antibodies. This conversion is achieved by perfusion of the lungs with two enzymes FpGalNAc deacetylase and FpGalactosaminidase. Science Translational Medicine, 2022, Feb 16 14 (632)” the authors described a method for ex-vivo conversion of carbohydrate blood group-A (GalNAca1-3Galb1-4GlcNAc-R) antigen into “universal donor” blood group-O antigen (Fuca1-2Galb1-4 GlcNAc-R) on endothelial cells of human lungs. Ex vivo enzymatic treatment converts blood type A donor lungs into universal blood type lungs. This strategy has the potential to expand ABO-incompatible lung transplantation and lead to improvements in fairness of organ allocation. These results show that depletion of donor lung A-Ag can be achieved with EVLP treatment. The treatment of donor lungs minimized antibody binding, complement deposition, and antibody-mediated injury as compared with control lungs. An ABO-incompatible transplant was then simulated with an ex vivo model of antibody-mediated rejection using ABO-O plasma as the surrogate for the recipient circulation using three donor lungs. No treatment-related acute lung toxicity was observed. EVLP of lungs with enzyme-containing perfusate removed over 97% of endothelial A-Ag within 4 hours. Baseline analyses of A-Ag in lungs revealed expression predominantly in the endothelial and epithelial cells. Eight ABO-A1 human lungs were then treated by EVLP. The enzymes removed greater than 99 and 90% A-Ag from RBCs and aortae, respectively, at concentrations as low as 1 μg/ml. First, the ability of these enzymes to remove A-Ag in organ perfusate solutions was examined on five human ABO-A1 RBC samples and three human aortae after static incubation. Here, we study the safety and preclinical efficacy of using these enzymes to remove A antigen (A-Ag) from human donor lungs using ex vivo lung perfusion (EVLP). The enzymes FpGalNAc deacetylase and FpGalactosaminidase, used in combination, have been described to effectively convert group A (ABO-A) red blood cells (RBCs) to group O (ABO-O). 
Donor organ allocation is dependent on ABO matching, restricting the opportunity for some patients to receive a life-saving transplant. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
